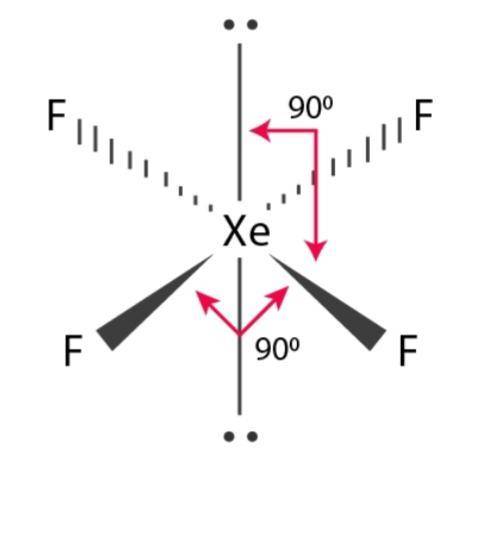
To complete its octet stability criteria each fluorine atom will share its 1 electron with the central xenon atom. This will make sure that the electron cloud is bent towards fluorine atoms thereby making sharing of electrons easier.Īs both xenon and fluorine in the XeF4 lewis structure are non-metals so there will be sharing of electrons. So xenon will be the central atom surrounded by 4 fluorine atoms. Between Xenon and fluorine, xenon is the least electronegative element and fluorine is the one with the highest electronegativity. So the total number of valence electrons are 8 + 7×4 = 36.Īfter finding the number of valence electrons participating in the XeF4 lewis structure, it is important to look out for the central atom. Similarly, fluorine (atomic number = 9 and electronic configuration = 2,7) belongs to group 7 of the periodic table and has 7 valence electrons. Xenon (Atomic number = 54 and electronic configuration = 2,8,18,18,8) belongs to group 18 of the periodic table and has 8 valence electrons. XeF4 lewis structure involves one atom of xenon and four fluorine atoms. Certain steps can be used to draw a perfect XeF4 Lewis structure. XeF4 lewis structure formation is the electron symbolism of the compound and confirms its stability and reliability. Another common use is in the xenon discharge tubes and flashbulbs used in cameras Another useful property of the XeF4 lewis structure is in space exploration where xenon is the main propellant. XeF4 lewis structure are used as ligands and coordinate with metal ions forming various fluorescent complexes. Due to this easy sublimation character XeF4, the lewis structure is sealed under vacuum in glass tubes.Īs such XeF4 lewis structure does not have major applications. XeF4 lewis structure exhibits the property of sublimation at room temperature. At -78 degrees celsius XeF4, lewis structure vapor pressure is negligible and at room temperature, it is approximately 3 mm. But now it has been confirmed that xenon and fluorine both can form stable XeF4 lewis structure under room temperature conditions.Įlaborating on XeF4 lewis structure properties and characteristics then it is a colorless solid. History and many academic research papers confirm that the first compound of xenon was not XeF4 lewis structure but Xe+ PtF6- as reported by scientist Bartlett. 
To achieve octet stability 4 fluorine atoms will share their 1 electron each with a Xenon atom thereby providing the desired XeF4 lewis structure.Įlaborating on XeF4 lewis structure then it is one of those handful compounds where noble gases participate in chemical bond formation. Similarly, fluorine belongs to group 17 of the periodic table and has 7 valence electrons. In the XeF4 lewis structure, xenon is a noble gas that has 8 valence electrons. XeF4 lewis structure and various properties associated with it are discussed in this article. Though noble gases remain in their elemental state, Xe is an anomaly. XeF4 lewis structure comprises noble gas xenon and the halogen atom named fluorine.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
